AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
What does the atomic theory state4/10/2024 There is another very useful kind of information contained in Ψ. But, it is known that even atoms of the same element can have different masses just like isotopes. Atomic theory, ancient philosophical speculation that all things can be accounted for by innumerable combinations of hard, small, indivisible particles (called atoms) of various sizes but of the same basic material or the modern scientific theory of matter according to which the chemical elements. The basics of the atomic theory are that atoms are the smallest units of chemical matter. Dalton states that atoms of a given element have exactly the same masses. Modern atomic theory is the best explanation atomic physicists have for how atoms look and behave. 
Soon after Schrödinger's proposal, his equation was solved for several atoms, and in each case the predicted energy levels agreed exactly with the observed spectra. Disadvantages of Dalton’s Atomic Theory: Dalton states that atoms are indivisible that they can be further divided into electron, proton and neutron. Once the form of is known, the allowed energies E of an atom can be predicted from the above equation. The details of how this is done are beyond the scope of this course, but the consequences of doing so are extremely important to us. Arrhenius theory, theory, introduced in 1887 by the Swedish scientist Svante Arrhenius, that acids are substances that dissociate in water to yield electrically charged atoms or molecules, called ions, one of which is a hydrogen ion (H + ), and that bases ionize in water to yield hydroxide ions (OH ). The atomic number is the number of protons in a nucleus. When the functional form of has been worked out, the Schrödinger equation is said to have been solved for a particular atomic system. All matter is composed of atoms atoms of the same element are the same, and atoms of different elements are different atoms combine in whole-number ratios to form compounds. As in any system composed of standing waves, certain boundary conditions must be applied, and these are also contained in Ψ the major ones are that the value of must approach zero as the distance from the nucleus approaches infinity, and that the function be continuous. Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 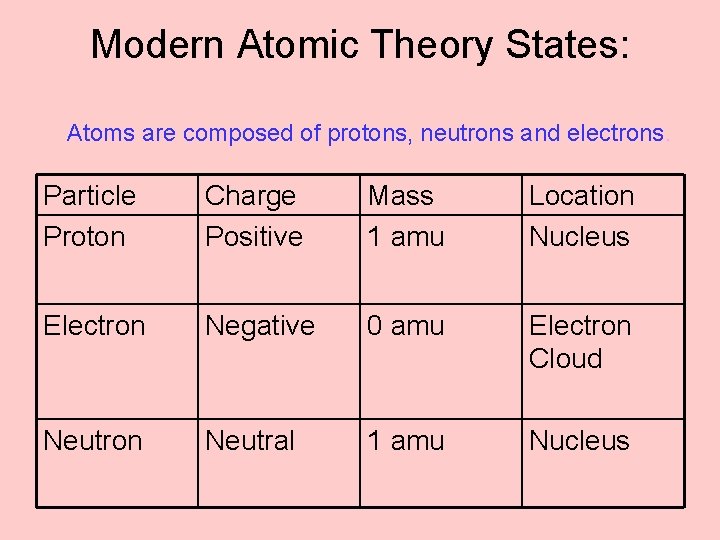
The rest consists of three basic types of subatomic particles: protons, neutrons, and electrons. Atoms can combine with other atoms to form molecules but cannot be divided into smaller parts by ordinary chemical processes. 4 ), is a fundamental concept that states that all elements are composed of atoms. atom, the basic building block of all matter and chemistry. As this names suggests, the value of Ψ is a function of location in space relative to that of the proton which is the source of the binding force acting on the electron. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5.4 1.5. 
How can such a simple-looking expression contain within it the quantum-mechanical description of an electron in an atom- and thus, by extension, of all matter? The catch, as you may well suspect, lies in discovering the correct form of Ψ, which is known as the wave function. Physical significance of the wavefunction
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
